Perform and quantify transwell-based live cell analysis of chemotaxis using the Celigo™
- Perform, image and quantify chemotaxis assays in microwell plates
- Automatic direct cell count and quantification of migrated cells
- Acquire Hi-res brightfield and fluorescent images of migrated cells
Perform and quantify transwell-based chemotaxis by direct cell count

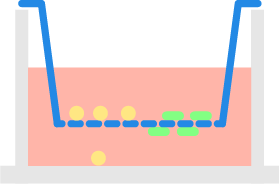
Suspension cells located inside the insert migrate through the porous membrane toward the chemoattractant from the bottom plate. The Celigo™ image cytometer is used to automatically count migrated cells in the bottom plate.
Imaging and quantification of MCP-mediated chemotaxis assay protocol
- Seed cells the transwell insert overnight
- Add regular cell media to the bottom of the wells
- At various concentrations add MCP (monocyte chemoattractant protein) to the bottom of the wells and incubate for 4 hours
- After four hours remove the transwell insert and image the migrated THP-1 cells
Images of dose-dependent MCP-mediated chemotaxis
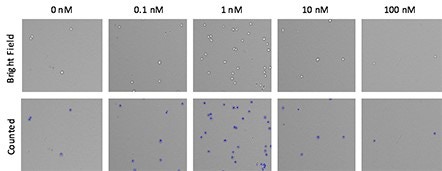
Brightfield images at each monocyte chemoattractant protein (MCP) dose were acquired and automatically quantified using the Celigo image cytometer.
Blue outlines indicate directly counted cells that migrated through the transwell insert into the bottom of the well.
Determination of viability of MCP- treated THP-1 cells

To determine whether MCP at various concentration had a negative effect on THP-1 cells, the cells were stained with propidium iodide (PI) and imaged.
Above are representative brightfield and fluorescent images show very few PI positive dead cells.
Viability data of MCP-treated THP-1 cells
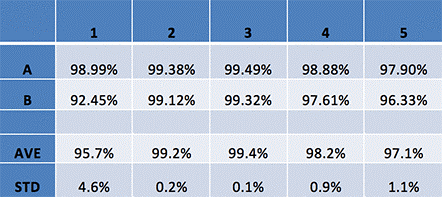
Based on the Celigo analyzed data we can conclude that MCP did not have any negative effects on THP-1 cells at any concentration.
The acquired averages and standard deviations across all samples showed minimal variation.
For research use only. Not for use in diagnostic procedures.




























