
Introduction
Since the quality of the cell sample is vital for potential downstream experiments, viability measurements are routinely performed in many laboratories. Choosing the correct method for conducting cell viability measurement is essential for obtaining consistent accurate results. Here we review a common method for measuring cell viability: Trypan Blue.
Using Trypan Blue to measure cell viability

Jurkat cells stained with Trypan Blue
One of the earliest and most common methods for measuring cell viability is the Trypan blue (TB) exclusion assay. Trypan blue is a ~960 Daltons molecule that is cell membrane impermeable and therefore only enters cells with compromised membranes. Upon entry into the cell, Trypan blue binds to intracellular proteins thereby rendering the cells a bluish color. The Trypan blue exclusion assay allows for direct identification and enumeration of live (unstained) and dead (blue) cells in a given population. Although Trypan blue has been used to determine cell viability for many years, it is not without its drawbacks. It is considered to be carcinogenic and must be handled with care and disposed of properly. Over time Trypan blue naturally forms dye aggregates and crystals. It is therefore recommended that TB is filtered using a 0.2 micron filter prior to use. Finally, multiple publications have observed that Trypan blue viability measurements in samples that are lower than 70 % viable show higher measured cell viability when compared to fluorescent based detection methods1,2. Data suggests that conducting viability measurements with Trypan blue on samples with low viability may not be optimal. We recommend that the Trypan blue assay is ideally used for cultured cell lines and purified/isolated cell samples with viabilities greater than 70%.
Trypan Blue protocol: sample preparation and analysis
Simple, user-friendly Cellometer procedure
Using Cellometer Auto 2000 and K2

1. Pipette 20µl 2. Insert slide 3. Select assay click count 4. Results in 30 seconds!
Trypan Blue protocol
- Dilute the stock (0.4 %) with PBS to 0.2 %
- Filter the Trypan blue with 0.2 micron filter
- Mix the cell suspension at 1:1 with 0.2 % Trypan blue
- Load the counting chamber slide into the Cellometer and analyze
Viability measurement of purified primary cell lines and cell culture using Trypan Blue
The micrographs displayed below represent cultured cells and purified primary cells that have been stained with Trypan blue and analyzed on the Cellometer instruments. The dark cells delineated with red circles represent dead Trypan blue positive cells. Cells with bright centers are considered live.
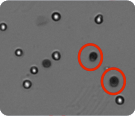
Using Cellometer Auto 2000 and K2
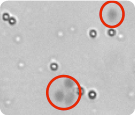
Splenocytes imaged by Cellometer Auto 2000
Conclusions & Cellometer selection guide
The Cellometer line of instruments can quickly obtain the viability of the cell sample by Trypan blue or fluorescent methods. Additionally, the system automatically reports an accurate cell count, concentration, and cell size in a single 20 µl assay. The fluorescent based Cellometer instruments (Auto 2000, Spectrum and K2) also allow researcher to count the total number of nucleated cells in a fresh clinical sample without the need to lyse red blood cells. The table below outlines the recommended Cellometer instrument and assay use for each selected application.
| Cellometer Auto T4 | Cellometer Ascend | |
|---|---|---|
| Purified cells – no debris | TB | TB or AOPI |
| Isolated MNCs – without lysing RBCs | NR | AOPI |
| Fresh BM, CB, WB, MNCs w/ lysing of RBCs | NR | AOPI |
| Fresh BM, CB, WB without lysing | NR | AOPI |
| Frozen BM, CB, WB | NR | AOPI |
| Digested tumors and BAL with tissue/cellular debris | NR | AOPI |
AOPI = Acridine Orange/Prodidium Iodide, TB = Trypan Blue, NR = not recommended, BM = Bone Marrow, CB = Cord Blood, WB = Whole Blood, MNCs = Mononuclear cells
For research use only. Not for use in diagnostic procedures.




























